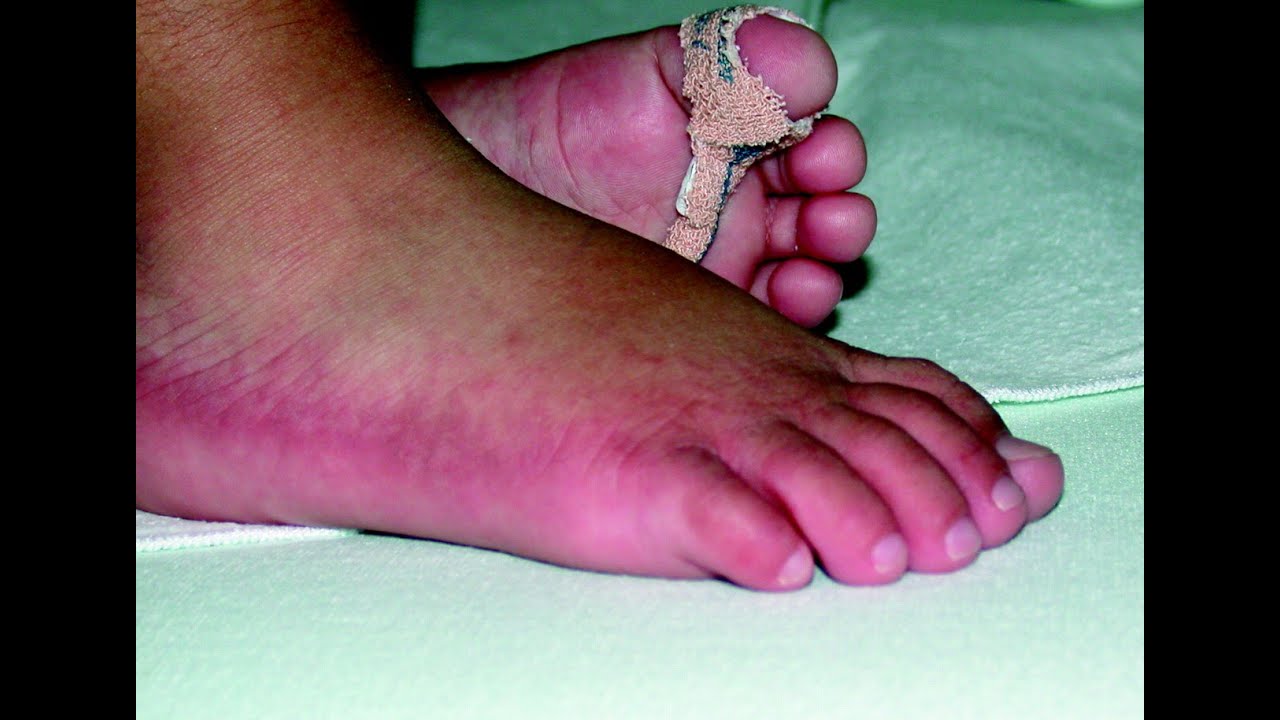
(4) In what must be one of the most awful ironies in science history, Wetterhahn was an expert on how toxic metals are able to penetrate cells. are sucked into vents and expelled outside the facility. They operate by negative pressure, so any fumes, vapors, etc. (3) Fume hoods are essential safety equipment in all labs. (2) "Sweet-smelling" may not sound too bad, but by the time someone has inhaled enough to detect the scent, he or she has already been exposed to dangerous levels of the chemical. They are usually very chemically reactive, which makes them useful reagents for chemical transformations. The properties of organometallic compounds are very different from organic (carbon-containing) chemicals. NOTES: (1) Organometallic chemistry is a subset of organic chemistry in which metals are bound directly to carbon. If you are interested in more detail on this horrific accident, you can find a good summary here. And, of course, dimethylmercury - the chemical that killed Professor Wetterhahn is an organomercury compound, and is, of course, far worse than methylmercury. That was, until it was discovered that certain bacteria in the water converted the mercury sulfate into methylmercury, and this ended up introducing a considerable amount of mercury into the environment. Since mercury sulfate is so insoluble in water, it was assumed (many years ago) that disposing of the stuff in lakes/rivers etc. wouldn't cause problems. 
The most common of these is methylmercury. When mercury is chemically bound to carbon it is readily absorbed into the blood, which makes organomercury compounds far more toxic than the other forms. Organomercury compounds: Of all the forms of mercury, these are the worst.Mercury sulfate and mercury iodide are essentially insoluble in water (7), so they tend to pass through the digestive tract without being absorbed, which makes them less toxic (but not non-toxic) than soluble mercury salts, such as mercury nitrate. Mercury salts are more toxic (orally) than mercury metal, but there is a wide range of toxicity, which depends mostly on water solubility. Mercury salts: These are chemical derivatives of elemental mercury that are formed when it is reacted with certain chemicals, such as strong acids.Mercury vapor is dangerous, since it is absorbed from the lungs into the blood and distributed throughout the body. 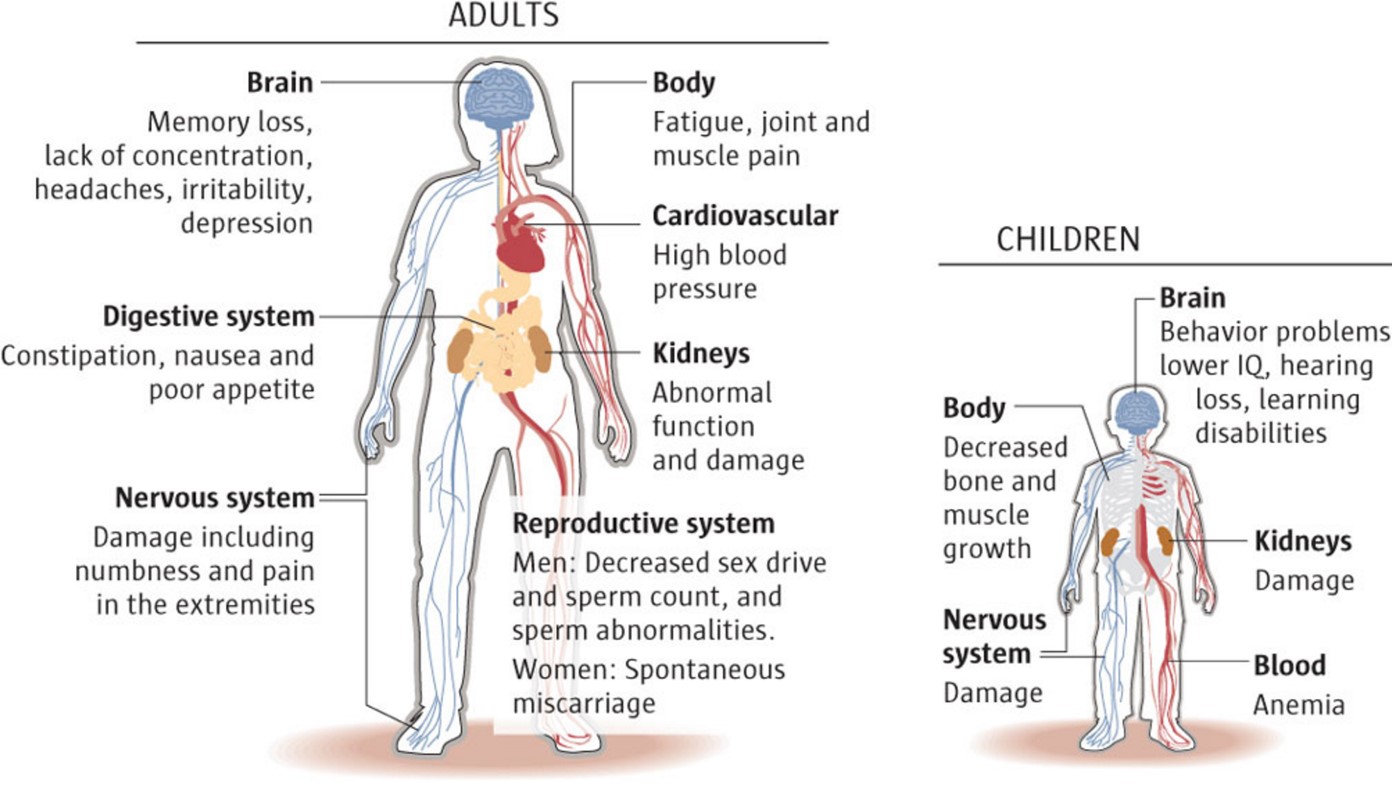
While ingestion of metallic mercury is relatively safe, the same cannot be said for its vapor. Although this "profession" no longer exists, it could be argued that it has evolved into a similar one: politics. But mercury was rare and valuable, so it had to be recovered from the stool (just in case you think *your* job sucks) and reused. This makes it a rather effective laxative, and was used as such in Asia thousands of years ago. Despite scare tactics used by "holistic dentists" in the 1980s, which caused many people to have all of their amalgam filling replaced, three large studies, including a 2014 Cochrane Review, which concluded, " insufficient evidence to support or refute any adverse effects associated with amalgam or composite restorations" have debunked that myth.ĭrinking mercury liquid itself is surprisingly safe (6), since it passes through the digestive tract without being absorbed. Silver-mercury amalgams have been used for dental fillings for almost 500 years. When mixed with other metals, it forms an amalgam.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
